 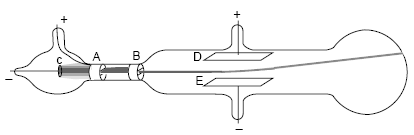
What evidence did Thompson have that led him to conclude the cathode ray was a stream of tiny negatively charged particles? Further experiments with magnets proved that the particles in the cathode ray also had mass. As a result, he reasoned that the particles in the cathode ray were negative. Thomson found that the path taken by the cathode ray could be bent towards a positive metal plate, and away from a negative metal plate. Why did he conclude that these particles were negatively charged? This showed that the charge carried by the cathode rays was negative. When the charged metal plates were introduced he found that the cathode rays bent toward the positive plate and away from the negative plate. Thomson set out to determine if the charge carried by the cathode rays was negative or positive. How did Thomson determine that the electrons have a negative charge?
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
